Science
Xenon Pharmaceuticals’ Seizure Treatment Shows Promising Trial Results
Xenon Pharmaceuticals announced on March 18, 2024, that its investigational treatment for a prevalent seizure disorder has demonstrated significant efficacy in reducing the frequency of seizures during a Phase 3 clinical trial. This outcome marks a critical milestone for the company, as the results surpassed those seen in its previous mid-stage study.
The trial involved participants suffering from a specific type of seizure disorder, and the results indicated a notable decrease in seizure incidents compared to a placebo group. This advancement not only reinforces the potential of the drug, named azetukalner, but also highlights the urgent need for effective treatments in the growing population of patients affected by seizure disorders.
Future Steps for Xenon Pharmaceuticals
Following these promising results, Xenon Pharmaceuticals plans to submit a New Drug Application (NDA) to the Food and Drug Administration (FDA) in the third quarter of 2024. This step represents a critical move towards making azetukalner available to patients who currently have limited treatment options.
The company’s Chief Executive Officer, Simon Pimstone, expressed optimism about the trial’s outcomes, stating that the data reflects a strong treatment effect and aligns with their objectives to provide effective therapies. The success of azetukalner in clinical trials not only positions Xenon favorably in the pharmaceutical market but also underscores its commitment to addressing unmet medical needs.
Furthermore, the trial results will likely influence the broader landscape of treatment protocols for seizure disorders. As the need for innovative therapies continues to grow, the efficacy demonstrated by azetukalner may pave the way for new standards in patient care.
In conclusion, the results from this Phase 3 clinical trial signify a significant achievement for Xenon Pharmaceuticals and its investigational drug azetukalner. With the anticipated FDA submission in the coming months, the company is poised to make strides in the treatment of seizure disorders, potentially transforming the lives of many patients.
-

 Lifestyle10 months ago
Lifestyle10 months agoLibraries Challenge Rising E-Book Costs Amid Growing Demand
-

 Sports9 months ago
Sports9 months agoTyreek Hill Responds to Tua Tagovailoa’s Comments on Team Dynamics
-

 Sports9 months ago
Sports9 months agoLiverpool Secures Agreement to Sign Young Striker Will Wright
-

 Science8 months ago
Science8 months agoSan Francisco Hosts Unique Contest to Identify “Performative Males”
-
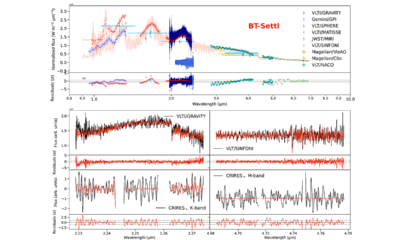
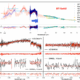 Science7 months ago
Science7 months agoAstronomers Unveil Insights from β Pictoris b Using GRAVITY
-

 Science6 months ago
Science6 months agoUniversity of Hawaiʻi Joins $25M AI Initiative to Monitor Disasters
-

 Lifestyle9 months ago
Lifestyle9 months agoSave Your Split Tomatoes: Expert Tips for Gardeners
-

 Sports9 months ago
Sports9 months agoElon Musk Sculpture Travels From Utah to Yosemite National Park
-

 Science9 months ago
Science9 months agoCalifornia Develops New Method to Monitor Toxic Algal Blooms
-

 Lifestyle9 months ago
Lifestyle9 months agoPrincess Beatrice’s Daughter Athena Joins Siblings at London Parade
-

 Sports9 months ago
Sports9 months agoBubba Wallace Makes History with Brickyard 400 Victory
-

 World9 months ago
World9 months agoWinter Storms Lash New South Wales with Snow, Flood Risks









